Overview: Pfizer Makes Strategic Leap with $43 Billion Seagen Acquisition

Basic Transaction Details:
| Acquiring Company | Pfizer Inc. |
| Target Company | Seagen Inc. |
| Per Share Equity Value | $229, at a premium of 35% |
| Transaction Size | $43 Billion |
| Announcement Date | 13th March 2023 |
| Financial Advisory |
Centerview Partners (Seagen) Guggenheim Securities (Pfizer) |
| Legal Advisory |
Sullivan & Cromwell (Seagen) Wachtell, Lipton, Rosen & Katz (Pfizer) |
Overview: Pfizer's $43 Billion Acquisition of Seagen
Pfizer (The Acquirer), an American pharmaceutical leader with a market capitalization of $226.8 billion, aims to acquire Seagen (The Target), a Washington-based biotech firm valued at $37.1 billion, for $43 billion at a 35% premium per share. The deal, anticipated to close by late 2023 or early 2024, is strategically set to accelerate Pfizer oncology pipeline by integrating Seagen's advanced Antibody-Drug Conjugate (ADC) technology. The merger aims to revolutionize cancer treatment by combining Seagen’s specialization in ADC technology with Pfizer's extensive R&D capabilities.
VeterinaRYAN's Analysis: A Medical Perspective of the Pfizer-Seagen Acquisition
“Pfizer is deploying its financial resources to advance the battle against cancer, a leading cause of death worldwide with a significant impact on public health,” said company CEO Albert Bourla
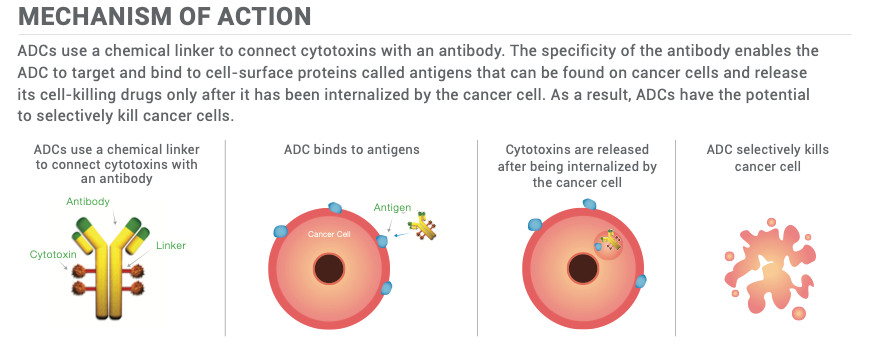
The recent acquisition of Seagen by Pfizer constitutes a paradigm shift in the arena of targeted oncological treatments. With the incorporation of Seagen's cutting-edge Antibody-Drug Conjugates (ADC) technology, Pfizer is strategically positioned to change the therapeutic index in oncology by obviating non-specific cytotoxicity and targeting tumour cells while sparing healthy ones. This mechanism could revolutionize the standard of treatment in oncology by reducing collateral damage to non-cancerous cells.
The acquisition also made Adcetris part of Pfizer's portfolio. Adcetris is an ADC that combines anti-CD30 antibodies with a cytotoxic payload, which specifically targets CD30-positive cancer cells, minimizing harm to healthy cells (as shown in the graph below). This FDA-approved drug has demonstrated clinical efficacy in Hodgkin's Lymphoma. It serves as a paradigmatic illustration of the therapeutic potential inherent in Antibody-Drug Conjugates (ADCs). Also, the inclusion of Adcetris not only enriches Pfizer's existing portfolio but also provides a strategically significant pipeline of new treatments. This is particularly important given that Pfizer's blockbuster arthritis medication, Enbrel, is facing challenges due to impending patent expiration.